Faster than ever, healthcare distribution in India is evolving; digital systems are at the center of this transformation. The demand for safe and interoperable systems has grown vital as more hospitals, clinics, and health tech businesses embrace digital processes. This explains why getting ABDM certification is no longer discretionary. It guarantees your platform satisfies the exacting standards for privacy, interoperability, and security, and runs perfectly within India’s National Digital Health Ecosystem.
Particularly in relation to compliance checks, sandbox testing, and cyber audits, many businesses find difficulties since the regulations appear complex and technical. This guide aims to make every requirement easier so that you can clearly grasp the ABDM eligibility standards, the ABDM certification process, and the precise ABDM compliance requirements you must meet by 2026. Useful links, examples, and expert advice to speed your approval also abound in the handbook.
You can always contact a specialist team like Qualysec, which assists healthcare firms at every stage of digital compliance, if you require personal direction or a partner to help you pass your ABDM audit the first time.
What Is ABDM Certification
Launched by the National Health Authority (NHA), the Ayushman Bharat Digital Mission (ABDM) seeks to establish a single digital health environment throughout India. Any digital health program has to get official ABDM permission if it wants to connect with this environment. This clearance verifies that your product safeguards patient information, employs the appropriate interoperability protocols, and meets national health data criteria. Getting this credential essentially demonstrates your system can securely share health records with other ABDM-registered systems.
India’s healthcare system has long been segmented; hence, this certificate is important, as each platform stores data in several formats. ABDM guarantees every system, regardless of who constructs it, speaks the same digital language.
Most medical institutions looking for digital integration need this certification. Among these are aggregators connecting many providers, diagnostic labs distributing digital reports, telemedicine applications enhancing consultations, and hospitals wanting interoperable EHR access. Early-stage firms, too, now see ABDM compliance as a main development objective since it boosts credibility.
Read our guide to healthcare security compliance requirements
Reasons ABDM Certification Still Counts In 2026
The foundation of India’s healthcare firms’ operations in 2026 will be ABDM integration. Interoperability is no longer discretionary, as adoption of digital health is growing dramatically. Healthcare platforms will stay isolated from national systems like the ABHA (Ayushman Bharat Health Account), Health Information Exchange, and provider databases without NDHM certification or ABDM authorization. This lack of integration directly influences both income potential and patient experience.
Organizations that forgo ABDM certification might find partner onboarding challenging as hospitals and diagnostic centers expect ABDM-ready digital systems more and more. Ignoring compliance by health tech companies especially raises cybersecurity hazards when dealing with sensitive patient information. Failing to fulfill these standards might cause fines, operational interruptions, or long-term reputational damage, given the government’s healthcare data security rules and more rigorous privacy regulations.
In essence, ABDM certification is more than simply a legal requirement. It indicates that your offering is safe, dependable, interoperable, and future-ready. Early attainment of this certification gives a competitive edge for organizations wanting to grow within India’s digital healthcare environment.
Criteria For ABDM Eligibility
Your company has to satisfy the ABDM eligibility standards, which specify the fundamental conditions for joining the digital health ecosystem, to start the approval process. These requirements guarantee that only pertinent and technically qualified platforms start the certification process.
1. Your Platform Has to Be Digital-Health Based
ABDM applies to any system that handles, stores, or shares health data. This covers platforms managing digital prescriptions, telemedicine applications, diagnostic information systems, and electronic medical record software. The intended use has to specifically include data management, patient care, or clinical procedures.
Examples of permitted product groups comprise
- Systems helping hospital operations are EMR/EHR
- Tools for teleconsultation control remote doctor-patient encounters
- Laboratory information systems that convert test results to a digital format
- Medicine ordering and prescription automation systems
The more clearly your platform’s medical use case is, the simpler your eligibility clearance.
Learn how medical device cybersecurity protects patient safety in healthcare
2. Interoperability must be supported by your system
Interoperability is central to the ABDM approach. Your platform must use approved APIs and established data formats. This guarantees easy transfer of medical records between several systems. Any departure from these patterns can cause sandbox denial, so technical teams must exactly adhere to ABDM schema rules.
3. Your system ought to adhere to the best security guidelines
Strict requirements exist in the areas of cybersecurity and privacy. Platforms managing health information need to show well-designed access control, strong encryption, secure authentication procedures, and audit logs. The ABDM cybersecurity assessment, which assesses whether your platform sufficiently safeguards patient data, makes these demands very important.
4. Registration on the ABDM Sandbox Portal is obligatory
Organizations have to sign up on the NHA Sandbox before any testing starts. This gateway lets teams test their processes in a regulated setting by means of ABDM APIs.
Requirements For Compliance with ABDM: You Must Fulfill
Compliance with ABDM, which is divided into four main categories, once you fulfill the requirements, will be the next action. These rules assist you in confirming whether your system can appropriately and safely interact with the digital health network of the nation.
I. Conformity Founded on Function
Your platform will run every required ABDM protocol thanks to functional compliance. This includes generating and verifying ABHA numbers, overseeing permission-based data sharing, allowing patient-controlled access, and disseminating physician or institution registry information. Every action has to follow the specified processes, operate perfectly, and give correct results. Testing helps to identify any contradictions that could cause retesting or feedback loops.
II. Concordance on the API level
Strict compliance of your APIs with ABDM’s encryption standards, reaction codes, and specification formats is required. Whether your API messages match the national schema, your platform’s request processing, and its response to errors all depend on the sandbox testing team. Rejection might arise from even minor variations like incorrect field names or unsupported data types.
III. Adherence with Security
Your platform protects patient data at every level via security compliance. This includes safe API contact, restricted access control, multilayer authentication, and encoded data storage. Among the faults found during the cybersecurity audit that might cause immediate certification delays are broken authentication, dangerous APIs, or bad session management.
IV. Observing Data Privacy
Privacy rules match the criteria under ABDM, including those in DISHA, the IT Act 2000, and worldwide criteria for patient data secrecy. Your platform must be defined by strict guidelines on data retention, anonymization, consent management, and access monitoring. Good documents let you know you are ready for compliance as well.
Want a secure foundation before entering ABDM sandbox testing? Explore Qualysec’s API security testing!
Step-by-step ABDM Certification Procedure For 2026
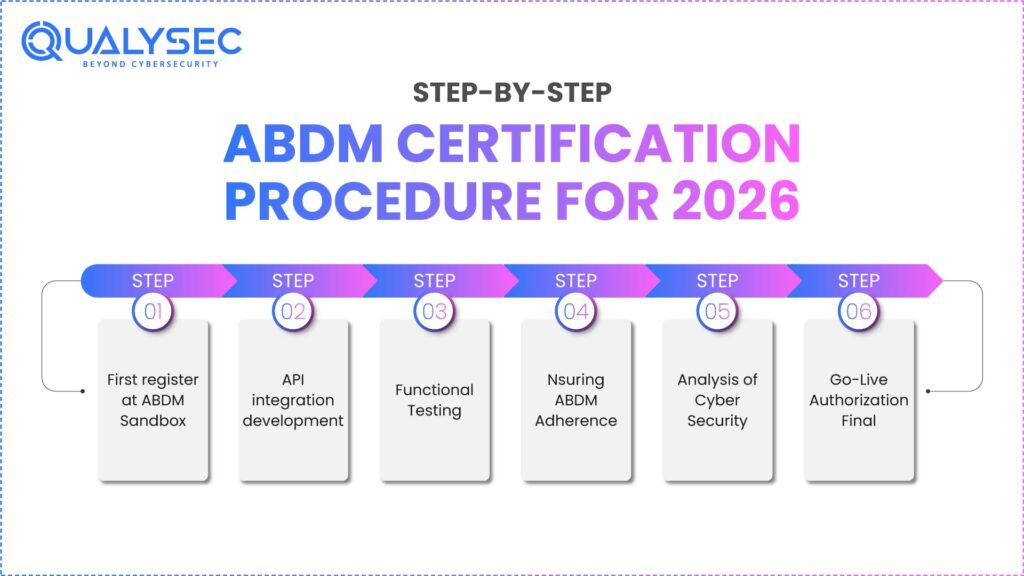
Six major phases make up the ABDM certification process. Every movement necessitates technical precision, appropriate paperwork, and ongoing contact with the NHA sandbox team.
Step 1: First, register at ABDM Sandbox
One needs product descriptions, API documentation, technical contact information, and detailed organizational data. ABDMs offer your onboarding credentials once accepted. This phase ensures your company is legitimate and your platform matches the context of digital health.
Step 2: API integration development
Currently, your development team links your platform with ABDM standards, including HIP (Health Information Provider), HIU (Health Information User), Gateway APIs, and consent management methods. Usually, this technical stage calls for several rounds of review before testing.
Step 3: Functional Testing
Your system would be proven to carry out all ABDM activities, including generating ABHA numbers, getting patient approval, retrieving electronic records, disclosing clinical information, and managing emergency access flows. Clearly written examination questions validate every situation.
Step 4: Ensuring ABDM Adherence
The sandbox team carefully examines API behavior, log clarity, security metadata, encryption techniques, and response consistency. To pass test cases, one has to resubmit for clearance and be corrected.
Step 5: Analysis of Cyber Security
Passing the ABDM cybersecurity assessment, which includes VAPT, API security testing, cloud audits, and OWASP-based testing, is vital. Organizations need to show great digital security before receiving final approval.
Step 6: Go-Live Authorization Final
Once all checks have been passed, ABDMs forward the certification letter and production credentials. This lets your platform legitimately participate in the digital health environment of the nation.
Learn how healthcare cybersecurity companies help protect patient data and critical healthcare systems
Full Timeline for ABDM Certification In India
| Stage | Estimated Duration |
| Sandbox registration | 2–5 days |
| API integration | 2–6 weeks |
| Functional testing | 1–3 weeks |
| Compliance testing | 1–2 weeks |
| Cybersecurity assessment | 1–2 weeks |
| Final approval | 1 week |
| Total Estimated Time | 6–12 weeks |
Want to evaluate your platform’s security before applying for ABDM? Start with Qualysec’s cloud security testing!
Is Vapt Required for ABDM Certification?
Yeah. Before certification, the ABDM cybersecurity evaluation calls for a thorough VAPT. VAPT confirms whether your cloud environment, APIs, backend infrastructure, and application are safe from cyberattacks. Health data is very sensitive, and weaknesses might have serious legal and operational repercussions.
Unsecured tokens, poor access constraints, incorrectly set up cloud storage, or unencrypted data transmission are among the problems found during the VAPT process. ABDM hopes these flaws will be addressed right away, and then a retest will follow. Many companies lose certification at this point since they underappreciate the depth of security examinations required.
Record your security policies, encryption protocols, and incident reaction plan to reduce risk. This gets your crew ready for the thorough examination ABDM will undertake.
Read our guide on choosing the right VAPT testing company
Frequent Causes Of Delays in ABDM Certification
Several problems might lengthen the certification process. Early knowledge of them will save you expensive delays:
1. Partial API integration
Many systems find difficulties in executing complete ABDM procedures. Sandbox rejections usually result from missing endpoints, inaccurate schemas, or inconsistent answers.
2. Wrong data mapping or schema disagreements
Validation failures are brought on by incorrect JSON structures or obsolete API versions. The most recent ABDM release notes have to be kept up to date by teams.
3. Security flaws found during VAPT
Weak authentication, risky APIs, or incorrectly set servers might prevent clearance until corrected. One of the most frequent reasons for postponement is this.
4. Vague or absent documentation
ABDM anticipates organized technical documentation. Back-and-forth with the sandbox reviewers usually follows from bad diagrams, unfinished flowcharts, and ambiguous logs.
5. Failure to make a case for data privacy regulations
ABDMs could demand significant modifications before permission if your data access or retention policies are lax.
Checklist: Your Needs Before Submission
Here is a detailed checklist to make sure your ABDM application moves ahead without incident:
- Well-documented arrangement of systems – Diagrams showing your platform’s data handling and transfer capability should be included.
- Technical specifications and API documentation– Verify that ABDM schemas and naming best practices match API endpoints.
- Figures showing data flow– Highlight patient-data movement throughout your internal and external systems.
- Security papers on policies– In corporate systems, include encryption methods, incident management plans, and access controls.
- Clean VAPT results– Your readiness for certification is revealed in a vulnerability-free report.
- Dedicated ABDM coordinator– Having a technical leader quickens correspondence with NHA and problem-solving.
Get a Free Sample Pentest Report

Internal Vs Abdm Security Expectations
| Security Area | Your Internal Standards | ABDM Security Expectations |
| Encryption | Basic encryption | Mandatory AES-256 + TLS 1.2/1.3 |
| User authentication | Password-based | Strong MFA and token validation |
| API security | Basic auth checks | Strict schema + digital signature |
| Cloud configuration | General setup | Hardened infrastructure + audits |
| Data access logs | Minimal logging | End-to-end traceable logs |
How Qualysec Can Help You Achieve ABDM Certification Faster
Having the correct support crew helping your compliance path makes obtaining ABDM certification much simpler. Qualysec distinguishes itself here by providing full support from documentation to security audit.
A. Complete ABDM Readiness Audit
Qualysec checks your system against ABDM eligibility criteria and finds flaws in data privacy, API readiness, and features. Before you even start sandbox testing, their specialists show you where improvement is necessary.
B. For ABDM Clearance, perform thorough security testing and VAPT
Qualysec does deep security testing across your application, APIs, infrastructure, and cloud environment since cybersecurity is among the most difficult aspects of certification. They guarantee your VAPT study is fit for presentation.
C. Hands-on assistance throughout sandboxing integration
Qualysec will step in to fix mistakes, verify schemas, and help your integration if your team has problems with functional flows or API mapping. This speeds up clearance and lowers rework.
D. Faster certification with knowledgeable direction
Qualysec helps you prevent ABDM delays by knowing the most often cited causes for them. Until your approval letter arrives, their staff assists you with the final submission, compliance document creation, and related requirements.
Conclusion
For healthcare institutions wanting to engage in India’s digital scene, ABDM certification is now among the most critical first moves. Early preparation and knowledge of every need help to make acceptance much more predictable, even if the process looks difficult initially. The certification process goes much more smoothly when your system passes the ABDM cybersecurity evaluation, meets all ABDM eligibility criteria, and submits full paperwork.
Qualysec gives everything you need to confidently achieve ABDM certification if you want a partner who can lead you through every step from readiness assessment to VAPT to final submission. Your platform draws closer to total compliance, enhanced security, and an easy interface with India’s digital health ecosystem with their professional assistance.
Schedule a call with Qualysec to discuss ABDM compliance requirements
Speak directly with Qualysec’s certified professionals to identify vulnerabilities before attackers do.
FAQs
Q1. What is ABDM Certification, and why is it required in India?
ABDM certification guarantees your digital health solution complies with national standards, is interoperable, and is safe. You cannot interact with the digital health services India is creating under NHA ABHA, provider registries, and health information exchanges without this permission.
Q2. Who is eligible to apply for ABDM certification?
Any digital platform that handles patient data, supports clinical workflows, or processes health records may benefit. This comprises hospitals, telemedicine systems, laboratories, pharmacies, EHR providers, and diagnostic applications.
Q3. How long does the ABDM compliance and testing process take?
The average certification period runs from six to twelve weeks. Organizations that lack documentation, fail functional testing, or do not pass the security evaluation cause delays.
Q4. Is cybersecurity or VAPT mandatory for ABDM certification?
Yes. ABDM ensures no vulnerabilities exist before approval by requiring a full security audit and VAPT.
Q5. What documents are required for completing ABDM approval?
System architecture diagrams, API documentation, data flow mapping, security policy papers, VAPT reports, and testing logs are all needed documentation.





















































































































































































































































































































































































































































































































































































































0 Comments