In 2026, cybersecurity is no longer a backseat concern for organizations operating in FDA-regulated environments. With the continued integration of medical devices, clinical trial platforms, and healthcare software and the increased reliance on software, regulators anticipate that companies will exercise greater control over cybersecurity threats that may compromise patient safety, data integrity, and system reliability. This has ensured that the FDA cybersecurity audit has become an important part of compliance and inspection preparedness.
Organizations under the jurisdiction of the U.S. Food and Drug Administration are required to indicate that cybersecurity is incorporated into their quality systems, risk management procedures, and system lifecycle. Such expectations extend beyond medical devices to clinical trial systems, cloud infrastructure, and third-party software used to support regulated activities.
The guide is designed to assist FDA-regulated businesses in planning and conducting cybersecurity audits in a risk-based and organized manner. It uses a pragmatic audit-planning model to help the FDA comply with cybersecurity requirements, mitigate inspection risk, and prepare organizations for 2026 inspections.
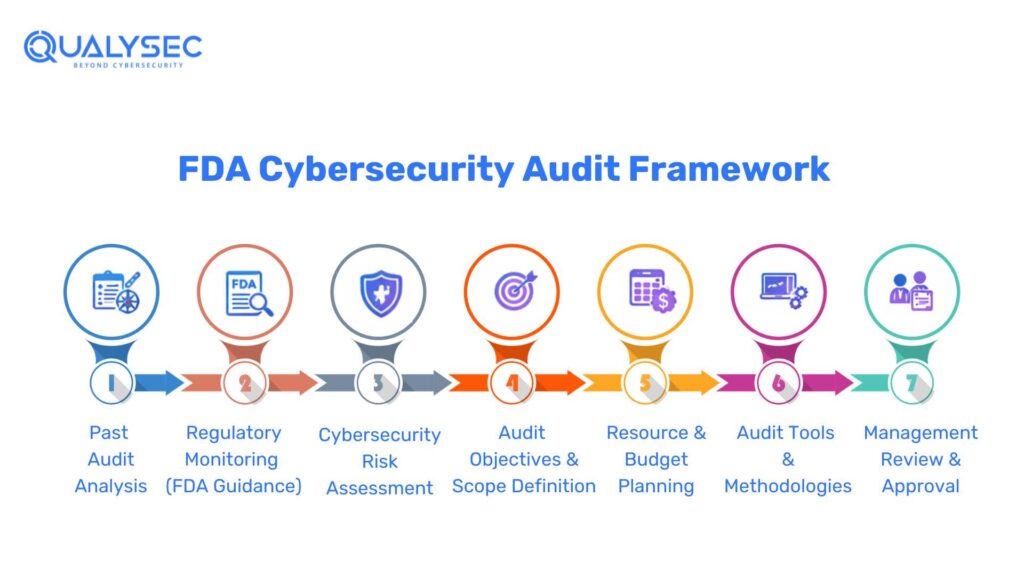
Review and Analyze Past Cybersecurity Audit Data for FDA Compliance
An efficient plan for an FDA cybersecurity audit starts with a transparent, objective analysis of previous audit performance. Cybersecurity audit data from the past will be insightful for identifying recurring issues, assessing the effectiveness of remedial measures, and highlighting areas that warrant greater attention to mitigate regulatory and patient safety risks.
Consolidate all cybersecurity-related audit records
The first step will be to consolidate all applicable cybersecurity documentation from the past audit cycle into a single accessible repository. This normally involves internal cybersecurity and quality system audit reports, third-party cybersecurity assessments and penetration testing recommendations, supplier and vendor cybersecurity audit reports, or any observations from inspections of software, data integrity, or system security. By consolidating such records, one will be sure that the analysis will be comprehensive and use relevant information.
Identify recurring cybersecurity findings
After consolidating the records, the organizations are expected to analyse the information and identify patterns and recurring findings. Problems that are observed across a series of audits, systems, or departments are most likely structural and not isolated. Aspects that may recur and be problematic include access control deficiencies, patching, incomplete documentation, and weaknesses in third-party oversight.
Evaluate the effectiveness of corrective and preventive action
Previous corrective and preventive measures must be examined to determine whether they were implemented as intended and whether they were effective in preventing the incidents. Raising of similar cybersecurity problems during the CAPA closure can indicate incomplete root cause analysis or insufficient remediation. This evaluation is especially crucial, as one expects to see repeat findings that can be considered during inspections.
Assess impact and risk concentration
Organizations should analyse areas with high concentrations of cybersecurity risks in their environment. This involves identifying medical devices or clinical trial systems whose findings are repeated, suppliers whose cybersecurity lapses are common, and systems that sustain high-risk or mission-critical regulated operations. These are the areas that would usually be of higher audit priority in the next cycle.
Use historical insights to justify audit priorities
Writing trends and lessons learnt from previous audits also help the organization clearly explain the audit priorities for the next cycle. This presumption of risk indicates that audit planning decisions are made using evidence-based arguments, which enhances FDA compliance in cybersecurity and minimizes the risk of finding the same issue during inspections.
Track and Assess FDA Cybersecurity Regulatory Changes in 2026
The FDA-regulated products, systems and their cybersecurity requirements are currently undergoing continuous changes, and regulatory awareness is an essential contribution to the audit planning. The examination of recent FDA cybersecurity guidance, the inspection trends, and the enforcement activity would serve to ensure that further audits are organized in accordance with the current expectations, but not with the past assumptions.
Monitor FDA cybersecurity guidance and inspection trends
New or updated FDA guidance on medical device cybersecurity and systems that are software-driven should be monitored proactively by organizations. Besides the official guidance, inspection patterns, warning notices, and enforcement measures related to software security and data integrity are also to be considered to get a clear picture of where the regulatory attention is growing.
Assess regulatory impact on systems and processes
Modifications in regulations must be considered in terms of their working impact on the current systems and processes. This also involves evaluating the impact of revised expectations on the medical device design controls, software development and testing, postmarket operations, and the cybersecurity of clinical trial systems. It is necessary to pay specific attention to the expectations of secure updates, vulnerability management, and data protection.
Update internal policies and procedures
Cybersecurity policies and standard operating procedures available internally need to be checked and changed to align with the modern FDA expectations. In the event of a changed requirement, the corresponding procedure must also be modified and documentation templates revised to reflect the kind of evidence that FDA inspectors are likely to demand.
Refresh audit criteria and checklists
New or revised regulatory expectations should be introduced or added to audit criteria and audit checklists. There should be more regulatory focus presented in areas that are explicitly covered as audit checkpoints. The tools should be updated and version controlled and communicated to audit teams to ensure that they are used consistently.
Assign responsibility for regulatory intelligence
The responsibility regarding monitoring FDA cybersecurity updates should be assigned clearly by the organizations. Keeping a regulatory change log that records changes identified, their impact and action needed will assist in ensuring accountability and traceability. The quality, regulatory, engineering, and IT stakeholders should receive regular updates on the regulatory changes.
Routine assessment of regulatory modifications helps make sure that the audit of cybersecurity is consistent with the existing expectations of the FDA inspection and offers a good ground upon which the risk assessment and audit planning practices will be conducted.
Conduct a Cybersecurity Risk Assessment for FDA-Regulated Medical and Clinical Systems
An effective cybersecurity audit program involves a targeted and justifiable FDA cybersecurity audit program on the basis of a cybersecurity risk assessment. This will help to target audit resources on the systems where the highest risk to patient safety, data integrity and regulatory compliance is observed, instead of spending evenly on low and high-risk areas.
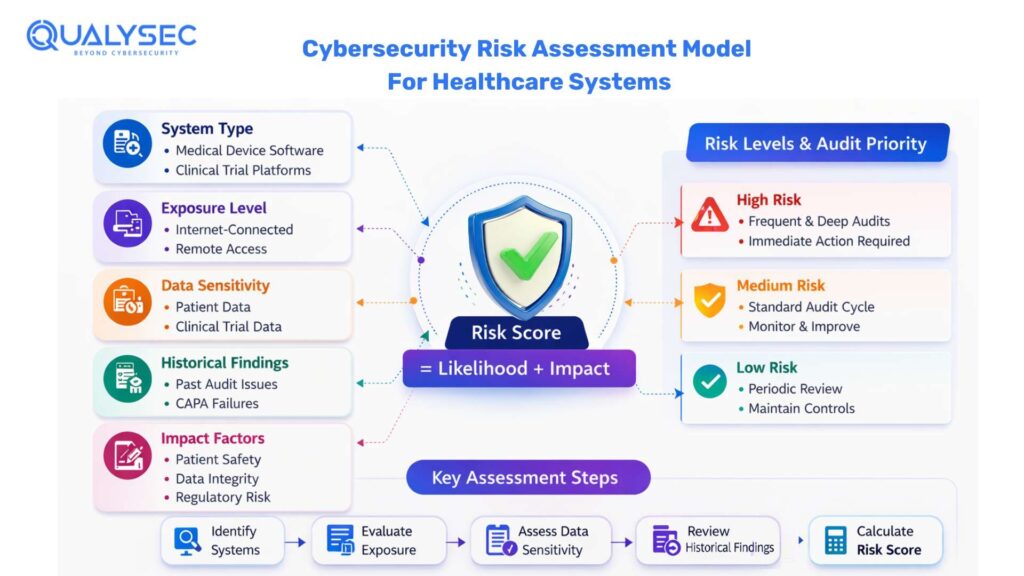
Define risk criteria and scoring methodology
To start with, organizations need to develop a uniform and documented mechanism for assessing cybersecurity risk. This involves the definition of the way the likelihood and impact are rated and the general level of risk. The categories of impacts must indicate the priorities of the regulated environment, namely, patient safety, product availability, data integrity, regulatory exposure, and business continuity. It is important that consistency at this stage is essential to defendable audit prioritization.
Identify all auditable systems and processes
The full list of the systems that may have an impact on the FDA-controlled products or data needs to be developed. This normally comprises medical device software and connectivity devices, clinical trial platforms, cloud computing infrastructure, quality system software, and third-party suppliers or service providers. Lack of complete system inventories usually introduces audit gaps and findings of inspection in the future.
Incorporate historical audit and inspection data
Risk scoring should be informed by the results of past cybersecurity audits, FDA inspections, and third-party assessments. Those with recurrent findings who have not been addressed by CAPAs and those whose findings have been noticed before should be classified as higher risks. Making risk decisions based on historical information supports the idea that these decisions are based on factual evidence and not theories.
Assess technical exposure and threat likelihood
The systems shall be assessed with regard to their exposure to cybersecurity threats. The internet availability, remote connectivity, updating systems, sensitivity of data, and dependency on third-party parts are some of the factors that determine the likelihood of threats, which should be recorded. Increased exposure usually justifies the increased depth and frequency of the audit.
Evaluate the potential impact of cybersecurity incidents
The organizations are supposed to gauge the impact that a cybersecurity attack would have on every system. This entails the influence on patient safety, integrity of the clinical trial, the performance of the product, regulatory requirements and continuity of operations. The higher the potential impact of the systems, the more they should be audited and supported.
Assign and document risk ratings
The impact and likelihood assessment needs to be united with the aim of providing a definite risk rating on each system or process. The reasons why the rating was made in a certain way shall be recorded in detail enough to facilitate discussions during inspection. Subjective or undocumented risk decision-making is not easily justifiable on audit or inspection.
Use risk ratings to drive audit priorities
The audit planning decisions should be directly related to risk ratings. The auditing of the high-risk systems must occur earlier in the cycle and be more extensive, whereas less frequent auditing of the less risky systems, but with written reasons, can be practised. This association indicates a systematic, recurrent method of audit planning.
An effectively documented cybersecurity risk assessment demonstrates that the planning of an audit is deliberate, regular, and knowledge-based. This action helps to have cybersecurity preparedness for FDA audits and makes organizations ready to establish clear audit goals and scope at the following phase.
Define Clear Objectives and Scope for an FDA Cybersecurity Audit
Once the cybersecurity risk assessment is done, the FDA-regulated companies should specify the goals and scope of every cybersecurity audit. It is a step to make audits focused, risk-based, and geared towards meaningful, inspection-ready results and not general or checklist-oriented results.
Establish specific and measurable audit objectives
The goals of every cybersecurity audit must also be stated in the form of clear objectives that outline the nature of what the audit is aimed at verifying. The objectives must provide details of the systems, controls or processes to examine and the desired result, e.g. ensuring that the vulnerability management process, access controls, secure update mechanisms, or incident response processes are effective. Clearly set tasks assist the auditors to remain focused and defendable when there are inspections.
Align audit objectives with risk assessment results
The cybersecurity risk assessment should be used to identify the risks that the audit objectives should reflect. The purposes of high-risk medical devices or clinical systems are to be reviewed and supported with stronger evidence and control effectiveness, in addition to a thorough review. Less risky systems can be associated with more focused goals, as long as the rationale is well-written and risk-based.
Include both compliance and effectiveness considerations
Besides the verification that cybersecurity controls are implemented, the audit objectives must also assess whether the cybersecurity controls are in practice. This difference will contribute to the fact that cybersecurity is not merely documented, but implemented, maintained and monitored in the long run.
Clearly define the audit scope
The scope of the audit must clearly indicate the products, systems, processes and locations covered by the audit. This could be in terms of particular models of medical devices, software applications, clinical trials platforms, cloud systems, or third-party providers. The definition of the scope allows not to be confused and contributes to the fact that the areas of high risk are not omitted accidentally.
Specify timeframes and lifecycle stages
The scope of the audit ought to determine the time frame which is being audited, say since the last audit or intercourse. It must also explain what lifecycle phases are encompassed, i.e. development, deployment, maintenance or post-market monitoring. The detail at this level promotes traceability and Inspection discourse.
Document exclusions and supporting rationale
The audit exclusions should be well-documented and justified using risks. Where questions arise in the course of an inspection, exclusions must be justified and reviewed on a regular basis as risks, systems, or changes in the regulations vary.
Formalize objectives and scope in an audit charter
The planning document or audit charter should be a formal document of audit objectives and scope. This forms a standard of reference to auditors and auditees, facilitates repeatable execution, and offers audit planning evidence inspection-ready.
A clear scope and objectives provide clear-cut audits on cyberspace security, which can be substantiated and are in tandem with the organizational risk priorities. This clarity forms the required basis of creating a realistic and effective audit schedule in the second step.
Resource Planning and Budgeting for FDA Cybersecurity Audits
Once the audit schedule has been developed, the regulated companies by the FDA should be able to demonstrate that they have the resources and funds needed to implement the cybersecurity audit program. The most effective audit plan may fail if it is not accompanied by an appropriate skill portfolio, capacity, and equipment. Resource planning is concerned with execution preparation instead of strategy.
Assess internal cybersecurity and audit capabilities
The first one is that the organization should start by assessing the level of expertise of internal teams to ensure that they can perform cybersecurity audits in FDA-controlled settings. This also involves an understanding of medical equipment cybersecurity, clinical trials security, regulatory expectations, and auditing based on risk. The shortfalls in skills, experience or availability should be determined at an early stage.
Identify specialized expertise requirements
Actions related to cybersecurity audits usually demand competencies that are not comparable to conventional auditing of quality or IT. The penetration testing, the review of secure software architecture, the vulnerability analysis and the third-party risk assessment areas might need specialized knowledge. Knowledge of these needs in advance enables the organization to make training or other support arrangements in advance instead of responding late to the audit cycle.
Estimate audit effort and workload
Every planned audit must be considered in terms of the time and effort needed to implement it in an appropriate manner. This involves preparing, auditing, documenting, reporting, and follow-ups. The relationship between the amount of effort required and the capacity demanded internally would be a way of assessing the reality of the workloads and their sustainability.
Plan for external support where appropriate
It is common for many FDA-regulated companies to outsource internal expertise, especially in high-risk medical devices of high risk or complex clinical systems. Outsourcing can also be used to offer external validation, technical richness and other capacities in times of peak audit duration. Prior planning of this support prevents waste of time and unexpected expenditures.
Budget for tools, testing, and supporting technology
The tools adopted in cybersecurity audits frequently include vulnerability scanners, penetration testing online platforms, an audit management system, and documentation repositories. The budget planning must consider licensing, maintenance and upgrades to be done to provide an inspection-ready evidence and audit traceability.
Allocate budget for training and capability development
In case internal teams are to complete or assist in cybersecurity audits, companies should invest in training and further education. This can involve Cybersecurity certifications, regulatory changes, or the FDA-oriented audit training to ensure that the teams are in line with the changing expectations.
Include contingency planning
Regulatory priorities and threats related to cybersecurity may shift at any time. This is due to the fact that by assigning contingency resources and budgets organizations are able to respond to incidents, newly discovered vulnerabilities, or urgent audit requirements without affecting the overall audit program.
Document and approve resource and budget plans
The audit planning must approve resource and budget decisions, which are to be documented. The management has clearly documented its support of the execution of the audit and has given evidence that is inspection-ready of governance and oversight.
Good resource planning and budgeting would make sure that cybersecurity audits are planned, as well as implemented effectively. This action equips the organizations to complete audit tools and methodologies in the second phase.
Finalize Tools and Methodologies for FDA Cybersecurity Audits
After resources and budgets are verified, controlled companies that are subject to FDA regulations should establish the tools and techniques for carrying out cybersecurity audits. This measure would make audits consistent, repeatable, and generate clear and defensible evidence that could be subject to FDA inspection.
Review and update cybersecurity audit procedures
Organizations would start by evaluating the existing audit practices to ensure that they are in line with the prevailing cybersecurity practices and regulatory standards. The procedures must specify how the audits are going to be planned, undertaken, documented and followed up. Any update that is a result of regulatory changes, internal improvement to the process, or information learnt during past audits must be included and version-controlled.
Standardize cybersecurity audit checklists
Audit checklists are important in providing consistency in audits. These checklists are to be revised in connection with the present-day FDA cybersecurity requirements and be individualized according to the type of audit, i.e., medical device audits, clinical trial system audits, and supplier audits. Standardization assists in guaranteeing that the essential requirements are examined continuously and flexibility in focusing on risks.
Define evidence collection and documentation requirements
The audits of cybersecurity should produce evidence of inspection readiness. Organizations are expected to have a clear definition of the evidence needed in each audit area and how they should record it. These are policies, risk assessment, validation documentation, security testing documentation, vulnerability reports, incident documentation, and CAPA documentation. Clarity levels minimize doubtfulness during auditing and inspections.
Establish consistent audit methodologies
The methods of the audit should outline the practice of the audits undertaken, such as review of documents, interviews, technical evidence analysis, and sampling. Regularized methodologies favor complete audits and enable the comparison of results among systems and audit periods.
Integrate root cause analysis and CAPA evaluation
The findings should be effectively analyzed using audit tools. This encompasses specific practices towards the root cause analysis and specific guidelines to decide when corrective and preventive measures are necessary. The combination of these factors will make audits meaningful and not merely superficial in their effect.
Leverage audit management and tracking tools
Where possible, the organizing and following up activities, audit schedules, audit findings and CAPAs should be handled via audit management systems or structured tracking systems. Having a centralized tracking system enhances visibility, accountability, and traceability in the FDA inspections.
Validate tools and train audit teams
The new tools and methodologies must be reviewed internally or piloted before they are used in audits. The audit teams should be trained on the revised procedures, checklists and documentation requirements to guarantee uniformity in their practice in all audits.
Concluding audit tools and methods ensure the cybersecurity audits are conducted in a systematic, repeatable and inspection-compliant fashion. This prepares the organizations for eventual review and approval of the cybersecurity audit program.
Final Review and Approval of the FDA Cybersecurity Audit Program
Once the cybersecurity audit tools and methodologies are developed, a formal review and approval of the entire cybersecurity audit plan should be performed by the FDA-regulated companies. This is to verify that the audit program will be coherent, sufficiently resourced, and prepared to run and give documented evidence of management control.
Conduct a comprehensive review of the audit program
The audit plan must be looked at as a holistic program and not as parts. The results of this review should ensure consistency in terms of risk assessment, audit objectives, scope, schedules, resources, and methodologies. The gaps, overlaps, and unrealistic assumptions must be detected and dealt with prior to the commencement of the audits.
Confirm alignment with quality and compliance objectives
The management ought to ensure that the audit plan facilitates wider quality and regulatory objectives. This will involve establishing that the risks of cybersecurity that may impact patient safety, data integrity, and product performance are properly discussed and ranked in the audit program.
Validate risk coverage and audit prioritization
Review process must ensure that the high-risk medical equipment, clinical systems and critical third-party dependencies are duly prioritized. Reasons why an audit is done, in which order and to what extent should be well recorded and justifiable in the audit discussion during inspection.
Review resource and budget commitments
The last look should also involve ensuring that there is enough manpower and other resources, including support and budget, to undertake the audit plan as intended. Any limitations or dependencies are to be recognised and dealt with in advance, prior to formal approval.
Obtain formal management approval
The cybersecurity audit plan, upon review, must be formally approved by relevant leadership, including quality, regulatory or executive management. Approval in writing shows that the organization is responsible and dedicated to compliance with cybersecurity.
Communicate the approved audit plan
The plan of the audit must be shared with the interested parties upon approval, such as audit teams, system owners, and supporting functions. Effective communication helps in getting the right expectations and plays a role in continuity.
An audited process of review and approval illustrates that cybersecurity audits are regulated, backed and aligned as per organizational priorities. This will position the organizations to use the external expertise in places where it is needed to enhance the effectiveness of audits further.
Strengthening FDA Cybersecurity Audit Readiness with Qualysec
With FDA cybersecurity requirements remaining immature, most of the FDA-regulated firms opt to enhance internal audit capacity with external cybersecurity services. Special assistance can enhance the quality of audit, offer independent assurance, and assist organizations to be ready to face more detailed and technically oriented FDA inspections.
Why external cybersecurity expertise adds value
In an FDA-controlled setting, cybersecurity audits demand a blend of regulatory knowledge, technical expertise, and audit discipline. Although internal teams can be of high quality or possess IT expertise, more complex medical device software, clinical trial platforms, and third-party dependencies can pose risks that can only be addressed with specialized cybersecurity expertise. Outside specialists offer a second opinion that will allow pinpointing the gaps and weaknesses that would not be noticed internally.
How Qualysec supports FDA cybersecurity audit readiness
Qualysec guides FDA-regulated companies by providing cybersecurity services which meet the FDA inspection expectation and audit evidence criteria. Their strategy is to produce audit-ready reports that can be utilized directly to support internal audits and inspections preparation, and regulatory discussions.
Some of the activities that Qualysec provides to support FDA cybersecurity audits include medical device and clinical system penetration testing, FDA-compliant cybersecurity risk assessment, and vulnerability analysis aligned with regulatory and quality system expectations. These services assist organizations to demonstrate that the cybersecurity controls are not only documented but also technically proven.
Strengthening audit evidence and inspection readiness
Evident and defendable cybersecurity effectiveness evidence is among the most prevalent difficulties when it comes to FDA inspections. External audit assessments that are conducted by independent specialists may reinforce documentation that is done by the auditors, by offering objective test results, structured risk analysis and well-organized reports that facilitate discussion and response to the inspections.
Supporting high-risk systems and third-party oversight
Medical devices that are high-risk, complex medical trial systems, and the critical third-party suppliers are especially good because of external knowledge. The independent testing and testing of these areas can assist organizations to give priorities on the remediation, limit the audit scope, and decrease the risk of inspection due to supplier and software dependencies.
Enabling continuous improvement beyond individual audits
In addition to individual audit activities, the external partners may assist in continuous improvement by finding weaknesses that are continually present in systems, refining audit processes, and enhancing compliance with FDA cybersecurity expectations. This assists organizations in switching to proactive risk-based cybersecurity governance rather than responding to compliance.
The exploitation of specialized cybersecurity skills is an opportunity that enables FDA-regulated businesses to improve audit quality, raise inspection preparedness, and gain more confidence in their cybersecurity positioning before FDA audits.
Conclusion
A cybersecurity audit in the FDA is more than a regulatory requirement in 2026. It is a direct measure of the safety of patients, clinical information, and the dependability of controlled mechanisms in an organization. FDA inspections are also becoming more focused on whether the cybersecurity controls are in place, but also on whether they are risk-based, are regularly applied, and have plausible technical evidence. Companies that apply cybersecurity auditing as a strategic, but not a compliance exercise, are in a better place to endure the pressure of inspection and minimize risk in the long run.
A value-based cybersecurity auditing program assists companies that are regulated by the FDA to find systemic vulnerabilities before they become critical, and provide compensation where it is most appropriate and show the ability to control complex medical and clinical settings. Combining historical audit experience, regulatory intelligence, structured risk assessments and disciplined audit executions will help the organizations change from a reactive response to inspection to a predictable, repeatable compliance. This would enhance the resilience of the operation and minimize the cost and disturbance of the last-minute remediation.
Partner with Qualysec to Strengthen FDA Cybersecurity Audit Readiness
Achieving successful cybersecurity audits is frequently associated with highly technical skills that extend beyond the normal quality of an IT role. Qualysec assists FDA-regulated companies to transform cybersecurity audits into a quantifiable business benefit by offering FDA-congruent risk audits, penetration testing, and audit-ready security testing.
Whether you are on the verge of an impending FDA inspection, dealing with high-risk medical devices or clinical trials systems, or enhancing third-party cybersecurity controls, Qualysec can help you to be audit-ready with independent, inspection-oriented skills.
Contact Qualysec to discuss how their cybersecurity services can help reduce inspection risk, improve regulatory confidence, and protect the systems that matter most.
Speak directly with Qualysec’s certified professionals to identify vulnerabilities before attackers do.
FAQs
Q: What is an FDA cybersecurity audit?
A: FDA cybersecurity audit examines the way the cybersecurity risks are identified, managed and controlled within the FDA-regulated systems. It concentrates on the safety of patients, data integrity and trustworthiness of the medical and clinical systems.
Q: Does the FDA require cybersecurity audits?
A: FDA does not require a standalone cybersecurity audit, though FDA inspection cybersecurity requirements require companies to show that they have effective cybersecurity risk management. Quality system audits, review of inspections and the FDA also tend to review cybersecurity.
Q: What systems are covered in an FDA cybersecurity audit?
A: FDA cybersecurity audit may include medical devices, clinical trial systems, cloud infrastructure, and third-party software to support regulated activities. Any system which affects the FDA-regulated products and data can be involved.
Q: What are common FDA cybersecurity audit findings?
A: The findings of the common FDA cybersecurity audit are weak vulnerability management, incomplete risk assessment, insufficient documentation and poor third-party oversight. Repeat discoveries usually include access control breaches and ineffective CAPAs.
Q: What documentation is needed for an FDA cybersecurity assessment?
A: FDA cybersecurity assessment usually needs risk analysis, policy, procedure, validation documentation, vulnerability report and CAPA documentation. FDA inspectors are not looking to see that controls are in place, but are operating.








































































































































































































































































































































































































































































































































































































































































































0 Comments