The filing of 510(k) submissions is not only among the most important but also the most difficult regulatory procedures that companies can undertake when introducing medical devices to the United States. The process of developing a product all the way to FDA approvals can be overwhelming, and is most often characterized by failures, misunderstandings, and unexpected delays. These hurdles are costly and frustrating to most companies, particularly during their first submission. This blog is a comprehensive overview of the five greatest 510k submission challenges companies encounter and how to overcome each one of them.
The 510(k) Pathway
A 510(k) filing is a premarket report to the FDA that proves that a new medical device is substantially equivalent to one that is already legally available in the United States. The process ensures safety and effectiveness while encouraging innovation, but the specifications remain daunting.
It’s not only about technical congruence. Regulatory science, evolving FDA requirements, and the complexity of device technology makes the process even more layered. Understanding the full scope and expectations of the FDA is fundamental for an efficient and successful submission.
Get the full guide on FDA 510(k) Guidance for Medical Devices.
Top Five 510k Submission Challenges
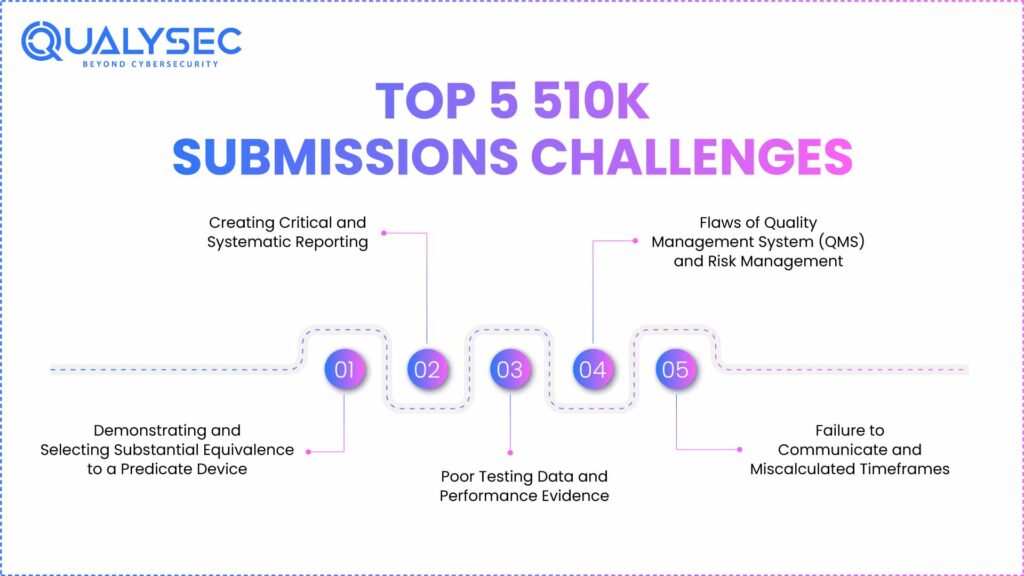
1. Demonstrating and Selecting Substantial Equivalence to a Predicate Device
Challenge –
The most important component of any 510(k) is proving that the new device is as safe and effective as a predicate device, which is a product that has already been cleared for marketing. Firms are not always capable of finding a suitable predicate or presenting powerful comparative data.
This can manifest as –
- Selection of an inappropriate predicate (not really comparable).
- Inability to outline similarities and differences.
- Missing performance data or risk analysis in the analysis.
How to Overcome?
- Research – Review FDA databases, and negotiate with regulatory gurus over which predicate is the best.
- Documentation – Be able to state similarities and differences, and support clinical/performance data behind claims.
- Regulatory Consultant Support – A recent 510(k)-experienced consultant can assist in pairing the product with the most suitable predicate and formatting the comparison.
Read also: What is FDA 510(k) Clearance? A Complete Guide.
2. Creating Critical and Systematic Reporting.
Challenge –
- Reviewers reject or withhold most replies simply because the papers are unfinished, inconsistent, or disorganized.
- Loss of critical test reports or descriptions of devices.
- Send conflicting information (e.g., contradicting technical specifications in other sections).
- Omit important justification when not including sections.
How to Overcome –
- Use the FDA Checklist – Use the most recent FDA Refuse to Accept (RTA) checklist and annotate it to show where you have fulfilled the requirements.
- Quality Check – Have someone on your team or a consultant go through it all and ensure that it is accurate and uniform.
- Standardized Templates – Prepare all documents using the standardized templates recommended by the FDA.
- Explain All Omissions – Where a section does not apply, give a written explanation as to the reason why it is blank.
- Routine Document Audits – Schedule periodic internal document audits throughout the development process to verify compliance, identify missing data, and ensure all content remains synchronized with evolving regulatory requirements.
3. Poor Testing Data and Performance Evidence
Challenge –
One of the most common reasons why 510(k) delays happen is that the submitted testing data, whether it be of biocompatibility or performance testing, is incomplete or badly supported.
Common pitfalls include –
- Incomplete protocols or raw data in the summary.
- Omission of all the risk and performance test data.
- Lacking evidence concerning important device operations (e.g., sterilization validation).
How to Overcome –
- Keep in Line with Current Standards – All tests and not only summaries, should be guided by FDA and consensus standards.
- Full Test Reports – Give clear results, thorough, and complete test protocols and rationales of all tests conducted.
- Early Planning – Add testing schedules (with repeat or follow-up testing) to project schedules to avoid end-of-the-day gaps in data.
- Employ Simulation Tools – Where feasible, supplement in vitro and clinical data with advanced modeling, simulation, and statistical analyses to offer robust performance evidence, especially for novel device features.
Must Read: FDA Cybersecurity Guidelines for Medical Devices 2025.
4. Flaws of Quality Management System (QMS) and Risk Management
Challenge –
Firms often fail in proper quality management and risk controls-both being under scrutiny of the FDA. The FDA does not only require a demonstration of a strong QMS, but it also requires documents of design and risk control measures across the device lifecycle.
Common issues –
- Absence of documentation of design control.
- Incomplete risk analysis.
- There is no structured or documented change management.
How to Overcome –
- Project Beginning – Introduce a known QMS (e.g., ISO 13485) at the beginning of the project and record all the activities.
- Design History Files (DHFs) – Measure design change, tests, and risk controls with full DHFs.
- Automated eDMS Tools – When thinking about digital management of documents, it is important to consider how quality records are to be arranged and gathered in order to be easily available.
- Cross-Training Personnel – See that the entire staff, including R&Ds and QA/QCs, is aware of their documentation and compliance tasks and duties.
5. Failure to Communicate and Miscalculated Timeframes
Challenge –
A communication breakdown with FDA reviewers is one of the most widespread, yet underrated issues; it is often exacerbated by miscalculating the entire review schedule.
Key pain points –
- Failure to expect questions from the reviewer.
- Fail to provide clear explanations that non-technical reviewers can understand.
- When, in reality, it may take six to nine months, including questions and data requests, to go through the review.
How to Overcome –
- Write Clean, Jargon-Free Submissions – To be reviewed–do not assume that all will understand your device. Write in simple and brief language and illustrations.
- Professional, Courteous Interactions – Speak to reviewers respectfully and promptly and cooperatively–even when there is disagreement.
- Anticipate and Prepare for Delays – Add to development timelines additional time to react to more requests for information and to engagement following submission.
- Pre-Submission Meetings – It is advisable to ask the FDA to hold pre-submission meetings (also known as Q-Sub meetings) to help them understand each other better and reduce unexpected results.
Need help overcoming FDA 510k submission challenges? Talk to our experts today.
A Stepwise 510(k) Success
Equipment Testing and Analysis
- Start by classifying the device appropriately according to the federal regulations. This involves ensuring what FDA product code is relevant, and whether a 510(k)-versus, say, a de novo or PMA is required.
- Premediated regulatory evaluation is useful to prevent channeling resources into the wrong direction.
- Examination of the competitive environment through the FDA 510(k) database of possible predicates and reading device summaries and clearance letters.
- You may want to ask for a pre-submission meeting to have the FDA concur on your intended regulatory plan and choice of predicate.
Predicate Device Selection
- The heart of any 510(k), and of the argument of substantial equivalence, is strong predicate device analysis.
- Compare predicted candidates to be used, technological characteristics, and risk profile. Defend the predicate of choice based on similarities and differences.
- Collect complete information about the predicate (public summaries, labeling, etc.) to argue and make the FDA see the point.
- Use predicates that were not recalled, taken in an enforcement action, or are technologically old.
- Create a comprehensive file, as recommended by the FDA. Provide cover letter, detailed table of contents, executive summary, description of device, labeling, predicate comparisons, performance data, risk analysis, sterilization, and shelf life data (as needed), and standards to be used.
- Preparation of support materials (examples – datasets, photos, test protocols) in apposite and easily navigable appendices or annexes.
- Lastly, check and complete the available RTA checklist and hand it in.
Gap Analysis and Review Inside
- Included with cybersecurity documentation of devices that include software, as per recent FDA requirements.
- Conduct a comprehensive internal audit, ideally using new eyes or other control consultants to spot discrepancies, pieces of information, or falsely presented arguments.
- The FDA can proactively avoid the line of questioning by its team members by conducting fake FDA reviewer sessions, i.e., role plays in which team members read off questions asked by the FDA.
- Close any loopholes or grey areas, justify why not, and only file after comparing with the latest version of the needs of eSTAR used by FDA.
Submission and Communication
- Electronically submit in the FDA eSTAR template and act proactively to maintain communication with the FDA during the review cycle.
- Prepare detailed, courteous responses to other information inquiries and keep detailed records of all communications.
- Whenever possible, designate a lead regulatory representative who serves as a contact point to facilitate queries and ensure a professional relationship with the FDA reviewer.
- Understand the feedback of the FDA and use the feedback to improve compliance performance by making successive submissions.
Let Qualysec be your US FDA Agent and make compliance easy.
Talk to our Cybersecurity Expert to discuss your specific needs and how we can help your business.
Conclusion
The process of submitting 510(k) applications is quite complicated and, in many ways, is not limited to technical innovation. Successful, timely FDA clearance is the art of preparation, clarity, and communication, which must begin with effective predicate selection, rigorous documentation, effective testing, compliance with QMS, and open communication with the FDA. By upstream preparing for these five 510k submission challenges, companies could have a much better chance to sail through the regulatory process seamlessly and finally gain access to safe and effective medical devices in the US market.
Qualysec helps you overcome 510k submission challenges, Book a meeting today.






































































































































































































































































































































































































































































































































































































































































































0 Comments