Key Takeaways
- Per CDSCO medical device software compliance requirements, the software should align with the October 2025 draft guidance and comply with MDR 2017.
- The rules now clearly distinguish between the SaMD (standalone apps) and SiMD (software integrated into physical hardware).
- The medical software is ranked from Class A to D, along with high-risk devices requiring Central Authority (CDSCO) approval.
- Manufacturers must provide the technical documentation with risk reports and special protocols for AI-based tools.
- The medical devices should go through penetration testing for software approval and selling in India.
Do you build or sell medical software or devices in India in 2026? Well, all those must comply with the CDSCO medical device software compliance to ensure safety from potential risks and threats.
In October 2025, the Central Drugs Standard Control Organisation (CDSCO) launched a guidance document on Medical Device Software. The guidelines were all about the clarity that SaMD developers, AI-based diagnostic companies, cloud-based health platforms, and mobile health app makers lacked.
CDSCO medical device software compliance is about the regulatory framework that sets the rules for all sorts of medical software and devices. In other words, this compliance helps to understand how medical software performs diagnosis, monitoring, prediction, and treatment practices. The framework ensures that such medical software or devices remain classified, licensed, and maintained in India according to the guidelines as per the Medical Devices Rules (MDR), 2017.
In this guide, let’s dig deep into what exactly CDSCO expects from risk classification, cybersecurity controls, documentation requirements, and post-market surveillance. Furthermore, we will also address how a reliable cybersecurity partner in 2026 can help your medical software fit into the compliance processes.
What Is CDSCO Medical Device Software Compliance?
If we check the Grand View Research – India Digital Health Market Report, the digital health market is thriving at a significant rate. In 2024, it was worth USD 14.50 billion, along with projected growth to USD 106.97 billion by 2033. This simply highlights that there are too many medical software, devices, and apps in the picture. That’s where the CDSCO medical device cybersecurity guidelines come into play, which govern the security, processing, and performance of the medical software.
CDSCO medical device software compliance involves processes and frameworks under which every medical software is scrutinized. The major parameters of software evaluation are based on its classification, validation, licensing, and monitoring under the Medical Devices Rules (MDR), 2017. As a high-level overview, there are two categories of medical software that are considered under this regulatory framework:
Software as a Medical Device (SaMD)
Individual or standalone software that performs any kind of work related to the medical field without the use of a hardware system is considered Software as a Medical Device (SaMD). Some of the SaMD examples include AI-powered radiology tools, ECG analysis, and clinical decision support systems.
Software in a Medical Device (SiMD)
Software in a Medical Device (SiMD) includes medical devices that contain inbuilt software systems. One of the key examples under SiMD can be firmware controlling an insulin pump or ventilator. Such software has a similar level of security as the hardware device.
All kinds of administrative or non-clinical software (that doesn’t involve any kind of medical screenings, diagnosis, or treatments) fall outside the CDSCO medical device cybersecurity guidelines.
Major Differences Between SaMD vs SiMD
| Assessed as part of the overall device cybersecurity | SaMD (Software as a Medical Device) | SiMD (Software in a Medical Device) |
| Definition | Standalone software performing a medical purpose independently | Software embedded within a physical medical device |
| Examples | AI radiology tools, ECG analysis apps, clinical decision support | Insulin pump firmware, ventilator control software, pacemaker software |
| Risk Classification | Independently classified (Class A–D) based on clinical impact | Inherits the risk class of the parent hardware device |
| Licensing Authority | Class A/B → State; Class C/D → CDSCO Central | Same as parent device’s classification authority |
| Post-Market Obligations | Standalone PMS plan required | PMS covered under parent device plan |
| Cybersecurity Scope | Full cybersecurity documentation required for networked/cloud SaMD | Assessed as part of overall device cybersecurity |
What Does CDSCO’s 2025 Draft Mean for Manufacturers?
As per the statistics, the global SaMD market for medical software is worth USD 34.05 billion in 2025. Because of this growth, the AI-based diagnostic tools now face much tougher scrutiny for cybersecurity and safety factors.
CDSCO released its Draft Guidance Document on Medical Device Software on October 21, 2025. It involves CDSCO software compliance requirements aligning with the nation’s regulatory frameworks. Moreover, the draft also covers the global scale standards (IMDRF, SaMD framework, and EU MDR) for medical software security and safety parameters.
The CDSCO’s Draft Guidance Document doesn’t create the law, but clarifies the role of existing MDR 2017 provisions. In simple words, it highlights how these regulations apply to the entire software lifecycle.
| Feature | Requirement/Change |
| Software Types | Clearly separates SaMD (standalone) from SiMD (embedded). |
| Risk Levels | Categorized from Class A (lowest) to Class D (highest). |
| Setup Process | Uses new templates and checklists to reduce paperwork delays. |
| Tracking Updates | Mandatory tracking for software updates and AI safety changes. |
| Security & Quality | Increasing expectations for cybersecurity controls and QMS in cloud-based software. |
| AI Protocols | AI tools must follow an Algorithm Change Protocol (ACP). |
| Licensing (A & B) | Managed by State Licensing Authorities. |
| Licensing (C & D) | Managed by Central Licensing Authority (CDSCO). |
| Submission Portal | Applications go through the CDSCO Portal or NSWS. |
SaMD Risk Classification Framework Under CDSCO Medical Device Software Compliance
CDSCO software validation medical device rules include a framework aligning with the risk identification-based model. Overall, the guidelines cover the clinical importance of software information and how serious illnesses can be addressed.
If the medical software is designed for people who lack clinical knowledge and is being used for diagnosis, it can be highly dangerous. Since the medical software works on its own with no professional medical doctor expertise, it needs to go through strict safety rules.
CDSCO SaMD Risk Classification Framework
| Risk Class | Clinical Significance | Healthcare Situation Severity | Examples | Licensing Authority |
| Class A | Low — treat or diagnose non-serious conditions | Non-serious | Wellness apps, basic health trackers | State Licensing Authority (SLA) |
| Class B | Moderate — inform clinical management | Serious but not critical | Chronic disease management apps, sleep monitors | State Licensing Authority (SLA) |
| Class C | High — drive clinical decisions | Serious or critical | AI diagnostic support, cancer screening tools | CDSCO Central Licensing Authority (CLA) |
| Class D | Highest — direct treatment or critical decisions | Life-threatening / critical | AI-guided surgical assistance, ICU decision tools | CDSCO Central Licensing Authority (CLA) |
What You Need to Submit for CDSCO Software Documentation Requirements?
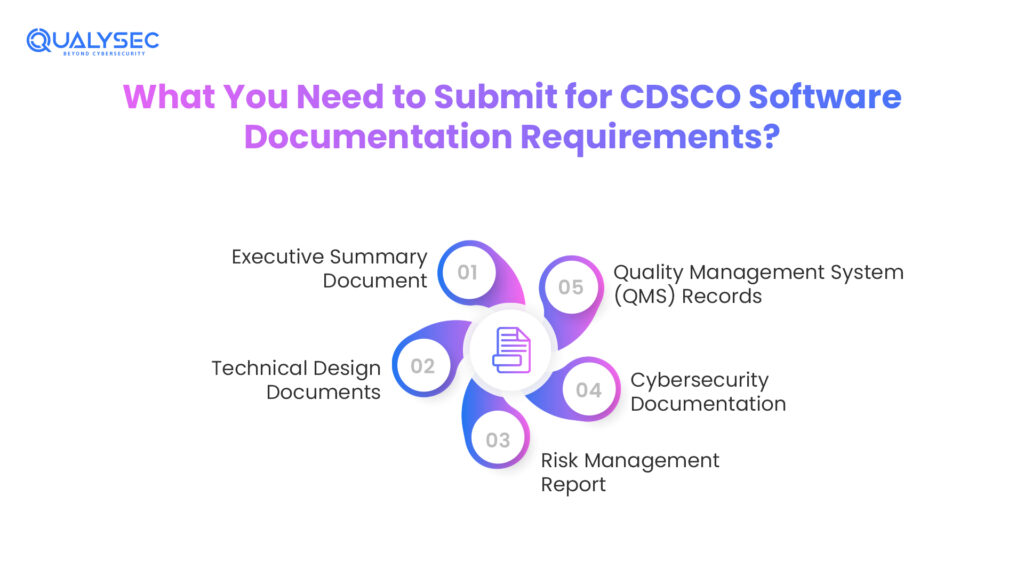
The 2025 Draft Guidance for CDSCO software compliance requirements offers the proper documentation checklist for SaMD manufacturers. No matter if you’re filing for the manufacturing license or import license, CDSCO expects a submission based on the medical software lifecycle.
Executive Summary Document
This includes the description, version history, and usage of the medical software. Further, SaMD regulatory compliance in India expects this document to cover the lifecycle model, logic, and clear terms. The summary document should also outline the input/output, change management, and more.
Technical Design Documents
As per the name, this document covers the technical architecture of the system and software applications. The medical software should add Software Development Lifecycle (SDLC) documentation along with validation records.
Risk Management Report
CDSCO software documentation requirements include risk identification, evaluation, and resolution as per the ISO 14971 standards. If the medical software uses AI/ML-based SaMD, the Algorithm Change Protocol (ACP) is highly mandatory.
Cybersecurity Documentation
This document covers the network connection details along with cybersecurity and data protection controls. Further, there should be security testing reports with penetration testing or more from the best cybersecurity company in India.
Quality Management System (QMS) Records
In certain cases, the CDSCO software compliance requirements might also need QMS framework evidence. There needs to be a post-market surveillance (PMS) plan along with compliant handling processes. The medical device manufacturers should share the software release certificate.
How Does Qualysec Help SaMD Manufacturers Meet CDSCO Compliance Requirements?
For medical software or device manufacturers, meeting CDSCO software compliance requirements is more than just submitting paperwork. As per the compliance guidelines, the medical software needs to prove that SaMD is secure, validated, and ready for the audit. This is exactly where Qualysec Technologies, the best cybersecurity company in India, adds more value.
Penetration Testing for SaMD Cybersecurity Compliance
CDSCO’s cybersecurity documentation requirements bind the medical software manufacturers to showcase their safety potential against cybersecurity attacks or threats. Our team of penetration testing experts conducts an end-to-end evaluation with a hybrid testing approach (a combination of automated and manual testing solutions).
We ensure that the testing remains in alignment with the healthcare application security parameters. Our team provides audit-ready reports that highlight the CDSCO’s disclosure requirements for cloud-based medical software.
VAPT and Security Risk Assessments
Qualysec performs Vulnerability Assessment and Penetration Testing (VAPT) dedicated to the medical device software requirements. This testing approach includes web interfaces, APIs, cloud networks, integrations, and more parameters.
VAPT and cybersecurity risk assessments by Qualysec help with the documentation related to risk and security management as per the CDSCO’s framework.
Compliance-Ready Security Reports
At Qualysec, we provide evidence-based cybersecurity reports that are helpful as per the CDSCO software documentation requirements. We double-check and ensure that the report findings remain mapped directly to the cybersecurity control areas as per the CDSCO’s 2025 Draft Guidance.
Ongoing Security Monitoring for Post-Market Obligations
CDSCO’s PMS requirements remain involved in the medical device manufacturer’s systems and processes. Qualysec’s managed security services assist SaMD manufacturers in maintaining continuous monitoring, faster vulnerability testing, and annual security retesting.
Are you planning to file with CDSCO in 2026 for your medical devices and software? Contact Qualysec to create security audit reports that fit with the CDSCO’s software documentation and guidelines.
Talk to our Cybersecurity Expert to discuss your specific needs and how we can help your business.
Qualysec Services Mapped to CDSCO Medical Device Software Compliance Requirements
| CDSCO Compliance Requirement | Qualysec Service | Deliverable |
| Cybersecurity documentation (networked/cloud SaMD) | Web Application & API Penetration Testing | Audit-ready pentest report with CDSCO-relevant findings |
| Security risk identification and mitigation evidence | VAPT — Vulnerability Assessment & Penetration Testing | Risk-mapped vulnerability report with remediation roadmap |
| Third-party component vulnerability tracking (SBOM) | Software Composition Analysis (SCA) | SBOM-aligned dependency vulnerability report |
| Post-market cybersecurity monitoring | Managed Security & Continuous Monitoring | Monthly security posture reports + incident alerts |
| Cloud infrastructure security (for SaaS-based SaMD) | Cloud Security Assessment (AWS/Azure/GCP) | Cloud compliance gap analysis against security baselines |
| Mobile app security (for mHealth SaMD) | Mobile Application Penetration Testing (iOS & Android) | Mobile-specific pentest report covering data storage, API, and auth |
Conclusion
Hence, CDSCO medical device software compliance highlights the regulations related to risks, licenses, and safety parameters altogether. For every manufacturer of medical devices or software in 2026, it is a necessity to stay in compliance throughout the software’s entire lifecycle.
Just like medical software performance, security is also equally important. The CDSCO software validation of medical devices considers cybersecurity as the key aspect to keep the patients safe. All the medical software companies need to show their software’s strength against security and other issues to get approval for selling in India.
Frequently Asked Questions (FAQs)
Q1.What is CDSCO software compliance for medical tools?
A: CDSCO medical device software compliance involves a set of regulations for software that helps doctors and medical practitioners find or treat illness. In simple words, any software that is used for medical or clinical practices in India must follow the Medical Devices Rules of 2017. Further, the new guidelines from October 2025 highlight the software classification, keeping it safe from hackers, and getting a license before selling.
Q2. Do the CDSCO auditors look at Software as a Medical Device?
A: Yes, they evaluate any medical app or program that works on its own for patient checkups. This includes the medical apps or AI that find diseases or track heart health. Such medical devices and software need to be tested for security and safety parameters before selling them in India.
Q3:What papers are needed to get software approved by CDSCO?
A: Yes, your medical software should comply with the CDSCO software documentation requirements from a safety point of view. This includes a summary of what medical software does, test results, and a plan for managing risks. If your medical software uses Artificial Intelligence (AI), you need to show how it changes over time with proper documentation and certifications.
Q4:How do manufacturers keep their medical software safe from hackers?
A: The manufacturers of medical software must document how they protect patient data from cyber risks and threats. Simply put, they need to show the use of passwords, secret codes, and a plan for fixing security loopholes. The best way to prove your software security standards is to get it tested for cybersecurity weaknesses.
Q5:Is penetration testing required for these CDSCO Medical Device Software Compliance regulations?
A: As per the new guide of 2025, your high-risk medical software that involves cloud systems and networks needs to be tested from cybersecurity parameters. CDSCO medical device software compliance expects both penetration and VAPT security test reports, which ensure that the software is safe to sell in India.















































































































































































































































































































































































































































































































































































































































































































0 Comments